Stem cell dishes
Stem cell dishes for organoids and spheroids
- outer dimensions 41x41mm
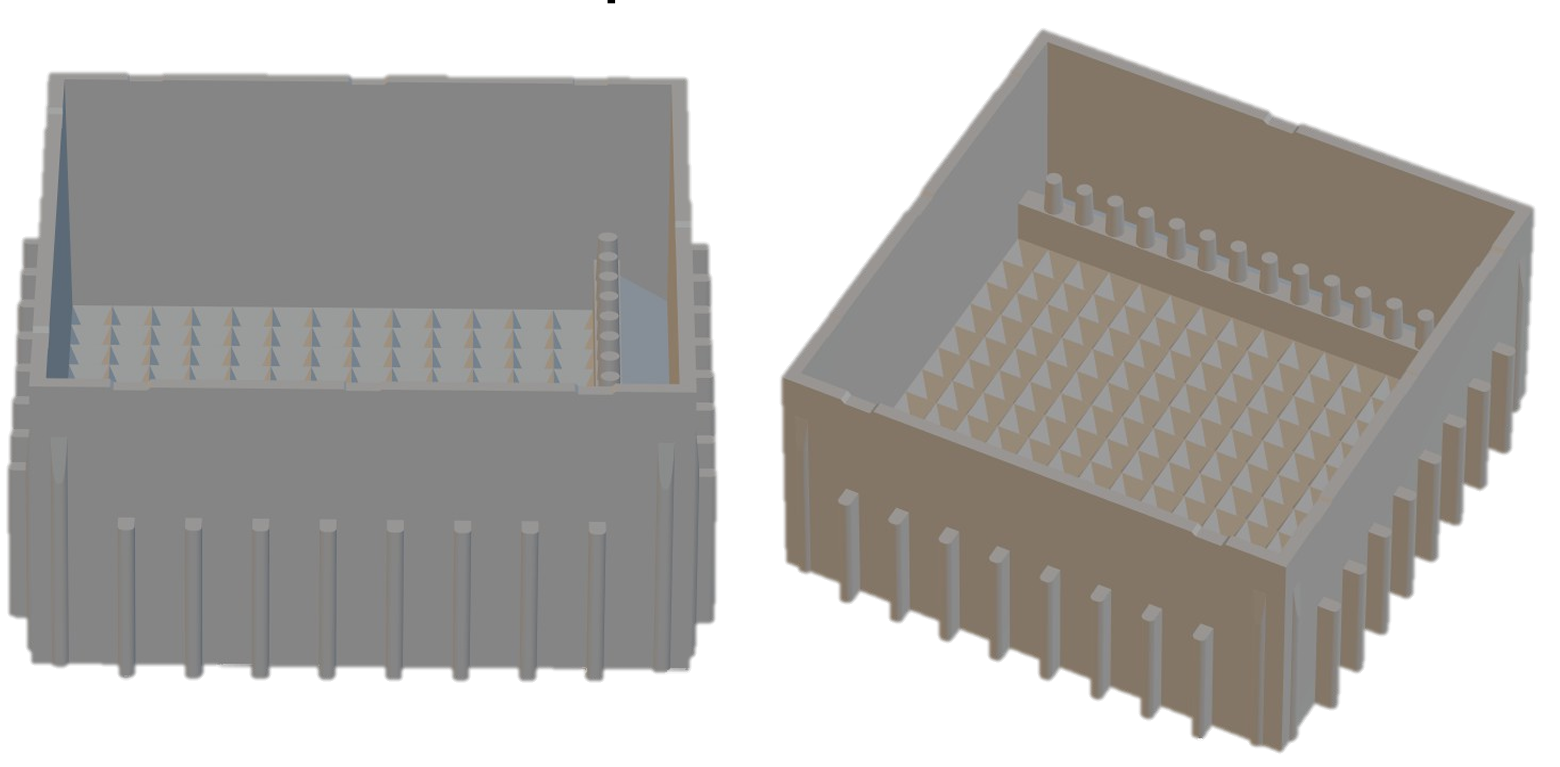
- 195 square microwells wells of 2x2mm
- 6 dishes can be combined (same footprint as a microplate; totally 1170 microwells)
- microwells have pyramidical bottoms so that cells accumulate in the center after seeding
- small reservoir behind vertical bars for an easy and gentle replacement of medium and aspiration (without spheroids being washed out of their microwells)
- poly-HEMA cotated medical grade polystyrene
- individually packed, and sterilized by gamma-irradiation
- transparent, square lid with 4 positions (rotate 90 degrees), defining various slit widths between dish and lid (enabling the choice of a specifc headspace refreshment rate)
- developped in collaboration with Dr. Hassan Rashidi (AuxiBio), a long-term specialist in liver and retinal stem cells

Protocols
1- Plate Priming
Prime the plate by adding 3 mL of PBS or DMEM. Remove any trapped air bubbles either by centrifuging at 800 × g or by gently pipetting up and down. If using DMEM, basal medium can be used with no supplements to keep the cost down.
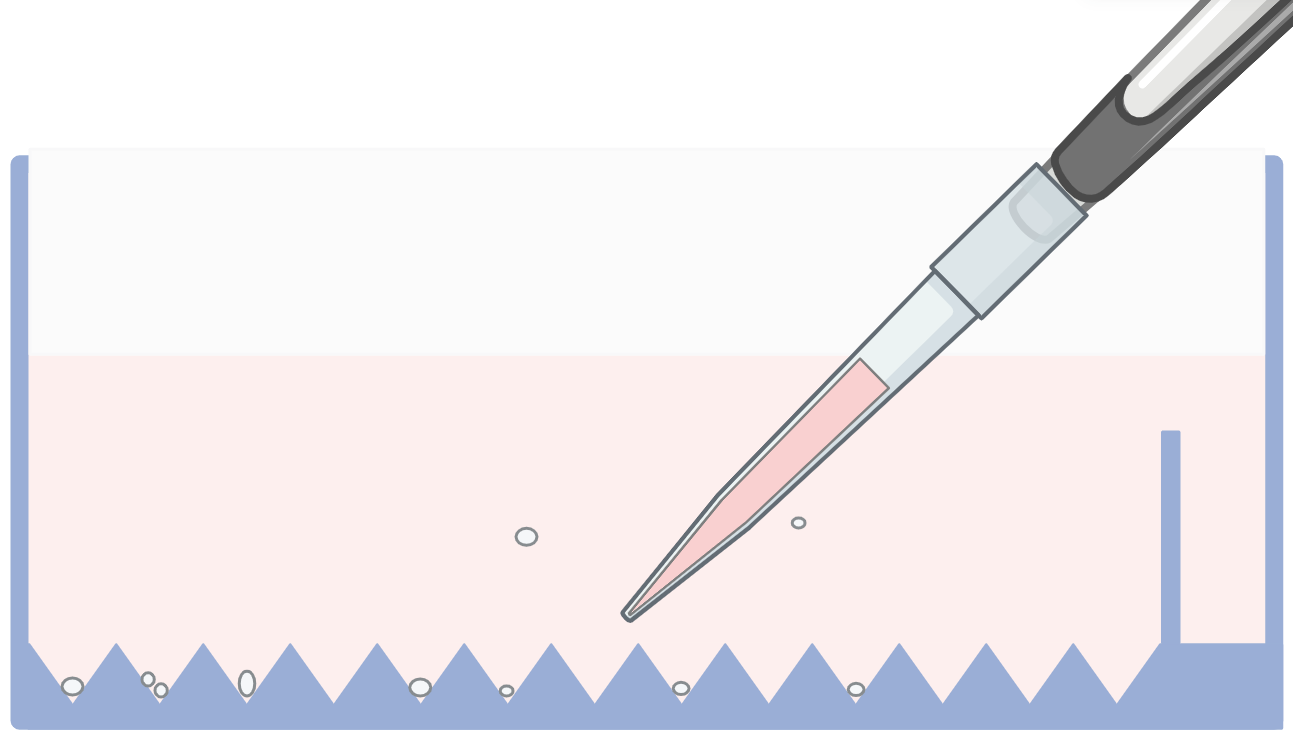
2- Medium Removal
Aspirate the PBS or culture medium. Tilt the plate toward one corner of the feeding compartment to remove as much liquid as possible.

3- Cell Preparation and Seeding
Dissociate cells into a single-cell suspension using an appropriate reagent. Calculate the required number of cells and resuspend them in 1.5–2.0 mL of culture medium.
For example, for 195 microwells requiring 5,000 cells per spheroid, prepare 975,000 viable cells (5,000 × 195) in 1.5–2.0 mL of medium. When working with human pluripotent stem cells, supplement the medium with ROCK inhibitor.
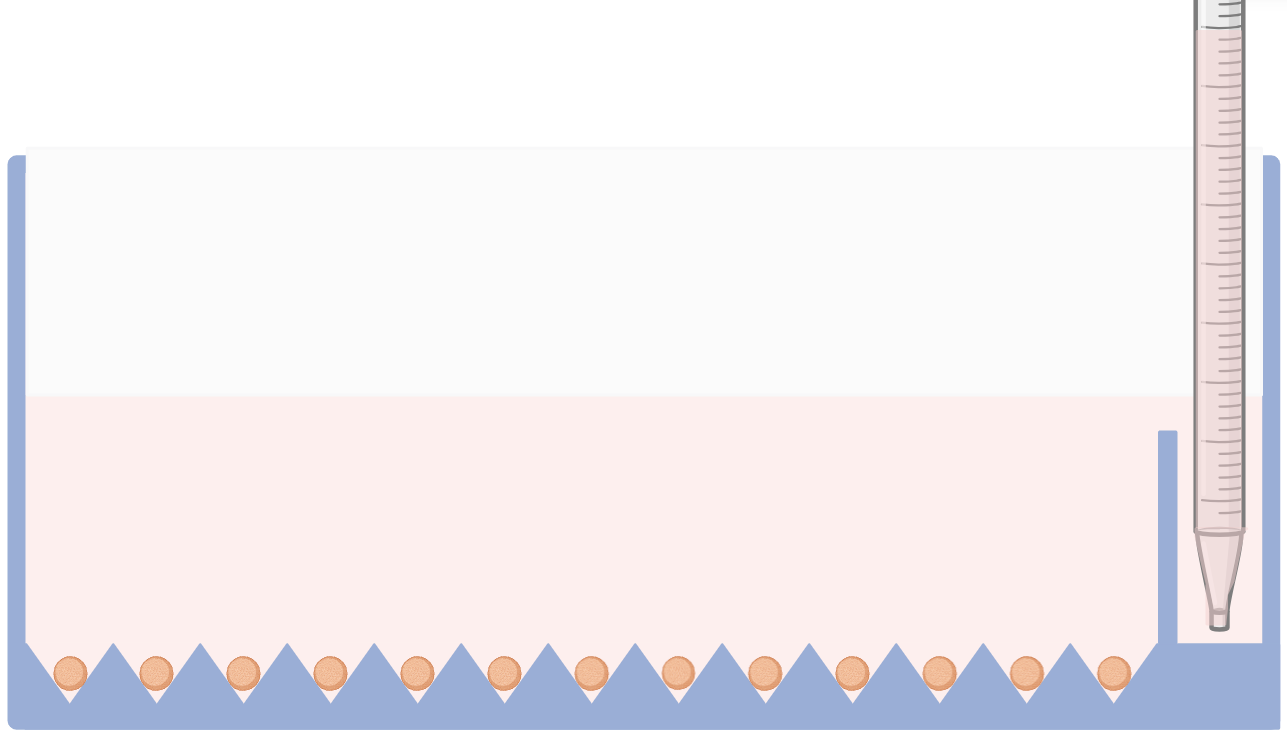
Gently add the cell suspension to microwell compartment ensuring no medium is added to feeding compartment.

4- Cell Aggregation
For optimal results, centrifuge the plate at 200 × g for 5 minutes. Alternatively, incubate the plate for 4 hours to allow cells to settle into the microwells and initiate spheroid formation.
5- Medium Adjustment (High Cell Density)
If seeding more than 5,000 cells per spheroid, gently add additional culture medium to the feeding compartment to a final volume of 3.0 mL.
6- Medium Exchange and Differentiation
Proceed with the desired differentiation protocol by aspirating spent medium from the feeding compartment and replacing it with fresh culture medium as required.
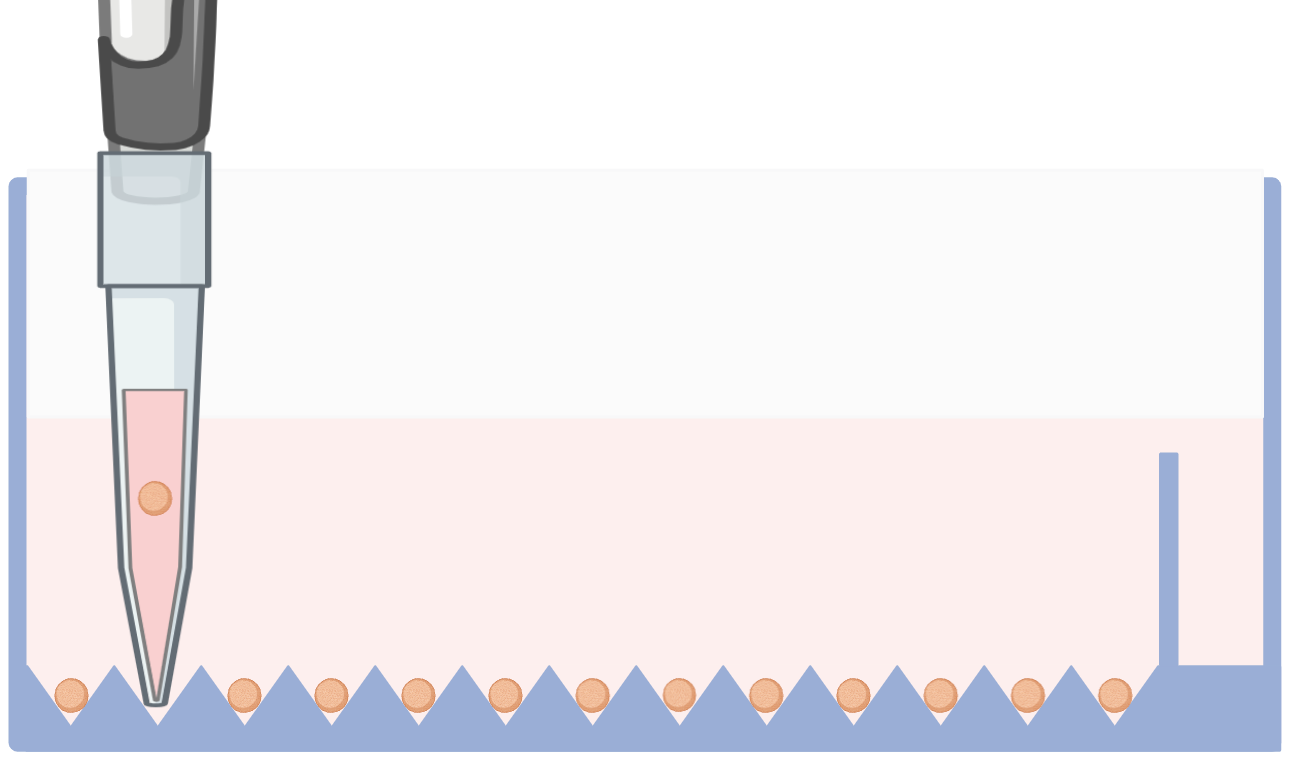
7- Spheroid Retrieval (Optional)
Individual spheroids can be retrieved without disturbing the remaining culture by placing a 1000 µL pipette tip directly over the selected microwell and gently aspirating the spheroid.
Contact us
The best way to contact us is to use the form below. However, you may also contact Wouter Duetz directly by e-mail. This e-mail address can be found on the "about us" page.
Optionally you may provide us with additional details about your project; culture type & duration, throughput and desired culture volumes.
We are happy to assist you. We generally reply within a single business day.
